Malaria
Malaria: origin and transmission
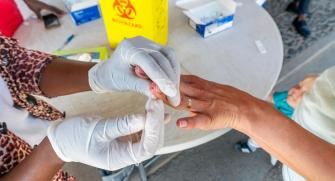
Malaria is caused by single-celled Plasmodium parasites that are transmitted to humans by mosquitoes. In 2022, there were an estimated 249 million malaria infections and 609 000 malaria deaths worldwide. Africa alone accounts for almost 94% of cases and more than 95% of deaths, and the disease continues to affect mostly children and pregnant women and children under 5 years old2.
In the early 2000s advances were made such as the roll-out of insecticide-treated bednets and the introduction of artemisinin-based combination therapy (ACT). Thanks to these interventions the number of deaths declined significantly between 2000 and 2015 and the incidence rate (cases per 1,000 inhabitants exposed to the risk of malaria) has dropped from 80 in 2000 to 57 in 20192, but they have recently plateaued.
In 2021, a major milestone was reached with the prequalification of the 1st malaria vaccine, RTS,S. The WHO recommends widespread use of the RTS,S/AS01 malaria vaccine in children in areas with moderate to high transmission of P. falciparum malaria. The vaccine has been shown to significantly reduce malaria morbidity and mortality in young children. 18 million doses have been allocated for distribution between 2023 and 2025 to 12 high-priority countries, with the first shipment of RSV,S arriving in Cameroon in November 2023. In October 2023, WHO prequalified a second safe and effective malaria vaccine, R21/Matrix-M.
Malaria has been the focus of Médecins Sans Frontières (MSF) for many years: 3 700 000 cases of malaria were treated by MSF in 2020.
Epicentre’s contributions have been focused on the diagnosis, treatment and prevention of malaria in MSF program settings in many different fields.At the end of 2024, Epicentre will start a clinical trial to compare two strategies for implementing vaccination with R21/MM.
Seasonal malaria chemoprevention (SMC) and other mass drug distributions

Epicentre accompanied MSF during the roll-out of seasonal malaria chemoprevention (SMC), a new strategy aimed at children under 5 in the Sahel. They are given prophylaxis drugs once a month during the rainy season when risk of malaria is high3 . Epicentre has carried out program coverage surveys of MSF-implemented SMC in Guinea-Bissau, Mali, Niger, and Chad, and provided advice on the continued roll-out of SMC4.
After the roll-out of the SMC strategy in Magaria, Niger, doubts arose about the program’s effectiveness, as the hospitals were still full of children with malaria. In 2016, Epicentre conducted a prospective case-control study to estimate the protective effectiveness of SMC against developing malaria. Because one of the concerns was that children were not correctly adhering to the 3-day course of treatment at home, the blood levels of the SMC drugs were checked. The main results showed that SMC is generally working well despite evidence of very poor adherence to the drug treatment.
In the Moïssala district in southern Chad, CPS has been implemented since 2013 by MSF in collaboration with national and local health authorities. Evaluation of the dynamics of malaria cases and hospitalisations over time and the expected impact of the strategy according to the implementation model adopted has led to the extension of the SMC to 5 distribution cycles in 2021, with the 1st cycle starting earlier in the transmission season. Further modelling work is underway to identify the most effective SMC implementation model and to assess the impact of other prevention strategies (such as vaccines) combined or integrated with SMC.
To protect vulnerable population, MSF organized a preventive treatment program for children <15 years in a refugee camp in northern Uganda. In 2015, intermittent preventive treatment of malaria (IPTc) was implemented in two refugee camps among children aged 6 months to 14 years. Three distributions of dihydroartemisinin-piperaquine (DP) were conducted 8 weeks apart.
Epicentre conducted a series of studies to describe this intervention. The knowledge gained should inform operational responses to crises of displaced persons both in Uganda and other high malaria transmission settings.

One of the risks of SMC, is that the malaria parasites may become resistant to the drugs used in the SMC strategy. The World Health Organization has recommended that routine monitoring of this takes place in areas where SMC is implemented. To do this, laboratory methods are used to look at the genes of the malaria parasites, and to see which ones might be resistant to the drugs. Epicentre has performed studies in Chad and Niger on this subject.
In Niger, the study showed that parasites with genotypes associated with the highest levels of resistance to amodiaquine and sulfadoxine-pyrimethamine are not yet common. However, it seems important to continue monitoring these resistance markers. This study also showed the heterogeneity of the parasites in a rather small area and the need to be cautious when extrapolating the results of surveys on molecular markers of resistance in a single site to deduce actions.
As early as 2014, Epicentre began a study of genetic markers of resistance to Moïssala, followed by others in 2021 and 2023. In 2021, the results showed the presence of the triple mutation in dhfr in most parasites (84% of cases), which had already been highlighted in 2014, and documented a clear increase in the quadruple mutation (triple dhfr + dhps437) from 28 to 41% of cases, pointing to a deterioration in the situation. The results for 2023 confirmed the upward trend observed previously. Quadruple mutations are present in 48% of samples, and quintuple mutations in 6%, compared with just under 3% in 2021.
Although estimates of markers associated with resistance cannot directly predict treatment failures, their assessments, especially their trends, provide indications as to whether the combination of sulfadoxine-pyrimethamine and amodiaquine remains an effective treatment for SMC. In order to assess resistance more accurately, in vivo studies of therapeutic efficacy are currently being developed.
MDA strategy in Ituri, DRC: coverage and impact on prevalence, morbidity, and mortality
For several years, MSF has been carrying out mass distributions of anti-malarial drugs (MDA) in part of the Angumu health zone, Ituri, in response to high malaria mortality. Since 2021, Epicentre has been evaluating their impact. First finding: MDA coverage is high at nearly 85%. The prevalence of malaria varies significantly between intervention zones: 62.4% in the routine zone without MDA or indoor residual spraying (IRS), 58.4% in zones with IRS, and 30.3% in the zone with MDA. In addition, the crude mortality rate and infant mortality rate were higher in the routine zones than in those using MDA.
Improving diagnostics
RDTs look for the presence of an antigen (a protein fragment) in the blood sample: most of these tests are for the HRP2 protein, which is specific to Plasmodium falciparum, and more rarely for lactate dehydrogenase (pLDH), which is present in all Plasmodium species. Tests for HRP2 have been preferred in areas where Plasmodium falciparum is predominant, due to their higher sensitivity. However, there are several limitations to this test. On the one hand, the antigen it detects remains present much longer than pLDH: an evaluation carried out by Epicentre in Uganda showed that the median time for an HRP2 test to become negative after effective treatment was 35 to 42 days, compared with 2 days for pLDH tests. So, in a high-transmission area, if a child receives effective malaria treatment after a positive RDT, then returns 28 days later with fever and a positive RDT, we can't know whether it's a new infection or whether there's another cause for his febrile illness. In areas of high transmission, HRP2 tests can therefore lead to overtreatment.
On the other hand, the performance of HRP2 tests is currently compromised by the emergence of Plasmodia with mutations that prevent the parasites from expressing the pfhrp2 and pfhrp3 genes, making them undetectable by conventional RDTs. First found in South America, these parasites, which do not express the pfhrp2 and pfhrp3 genes, are now found - albeit with very different prevalences depending on the country - in Asia, the Middle East and Central, East, South and West Africa.
An Epicentre study in collaboration with MSF is currently underway to assess the prevalence of these mutations in South Sudan.
Developing better treatment strategies
Field and clinical trials
Epicentre has performed and participated in several recent clinical trials related to the treatment of malaria.
- In Niger, one study evaluated three commonly-used ACTs - Artemisinin-based combination therapy - for the treatment of uncomplicated malaria, and found that all three had good effectiveness.
- The treatment of malaria in malnourished children can be challenging. In a clinical trial in Mali and Niger, artemether-lumefantrine was found to be equally effective in malnourished and non-malnourished children, but the levels of the drugs in the blood were significantly lower in malnourished children, possibly leaving them more vulnerable to reinfection9.
- Epicentre was one of 11 trial sites for the AQUAMAT trial, which showed that intravenous artesunate therapy reduced mortality from severe malaria in children by 22% compared to quinine treatment. This trial led to a change in WHO recommendations for the treatment of children with this extremely lethal form of malaria.
DeTACT to uncover new therapeutic approaches
Epicentre's Niger Centre was one of 14 sites - 8 countries in Africa and 5 in Asia - of the DeTACT project coordinated by MORU and funded by UKaid and the UK Foreign and Commonwealth Development Office (FCDO). It aimed to discover new therapeutic approaches against malaria. Indeed, resistance to new drugs combining an artemisinin derivative with one of the many anti-malarial molecules has started to appear in Asia and is now spreading in some regions of South East Asia. The DeTACT project is studying the efficacy, safety and tolerability of two artemisinin-based combination therapies (ACTs) using existing antimalarial drugs (artemether-lumefantrine + amodiaquine and artesunate-mefloquine + piperaquine).
In Niger, preliminary results favor a better post-treatment prophylactic effect with TACT compared with ACT and no signs of artemisinin resistance were observed. Further results will evaluate PCR-corrected efficacy, pharmacokinetics, pharmacodynamics and investigate the outcomes from the other study sites.