Research Center in Uganda
Research
Epicentre’s Uganda Research Centre conducts health related research aimed at influencing policy globally and relevant to Africa and Uganda in particular.
The Mbarara Research Centre laboratory in Uganda is GCLP (Good Clinical Laboratory Practice) accredited by the international and independent accreditation system, Qualogy. Studies are conducted according to Good Clinical Practice and Good Clinical and Laboratory Practice quality standards. Epicentre collaborates closely with the Mbarara University of Science and Technology (MUST), the Mbarara Regional Referral Hospital (MRRH), the Ministry of Health and other stakeholders in research, academia and the health sector.
The centre has an experienced team of medical doctors, nurses, social workers, statisticians, biologists, laboratory technicians, information technology scientists, logisticians, and administrators who ensure the quality of the research.
Epicentre research has contributed to public health policy changes including the seminal AQUAMAT trial that demonstrated that artesunate significantly reduces mortality in African children with severe malaria. Current projects continue to focus on improving diagnosis and therapies for infectious diseases such as tuberculosis, HIV, central nervous system infections, as well as vaccines trials on Ebola and Yellow fever.
With regard to tuberculosis, the center is involved in several large-scale projects. It is one of the sites of the TB-Speed study coordinated by Université de Bordeaux, which aims to reduce infant mortality due to tuberculosis. This infectious disease remains one of the main causes of death in children under 5 years of age and most of them die without having access to treatment because they are often not diagnosed. The goal of the TB-Speed study is to identify rapid, effective, and easy-to-use diagnostic tests to improve the diagnosis of TB in children.
The Mbarara Center is also collaborating in international treatment trials to simplify or reduce treatment regimens, which currently involve taking medication daily for 6 to 8 months. If treatment could be reduced to 3 or even 4 months, it would improve compliance and daily life for patients while limiting the emergence of drug resistance. After several previous studies, the international Rifashort trial is evaluating the efficacy and safety of a 4-month regimen using double and triple doses of rifampicin. The EDCTP and ANRS-funded DATURA trial is comparing the efficacy of more intensive initial treatment of TB with higher doses of the main antibiotics used, rifampicin and isoniazid, plus corticosteroids, in hospitalized adults and adolescents co-infected with HIV and TB versus standard treatment.
The Center also included the first patient in the DNDi-coordinated "LOLIPOP" study, which evaluated the 4-in-1 treatment for children with HIV in Uganda. This treatment combines four drugs needed to treat pediatric HIV in a single pellet-filled capsule that is easy to use, heat stable, tasteless, solid, and does not contain alcohol or inappropriate solvents.
Laboratory

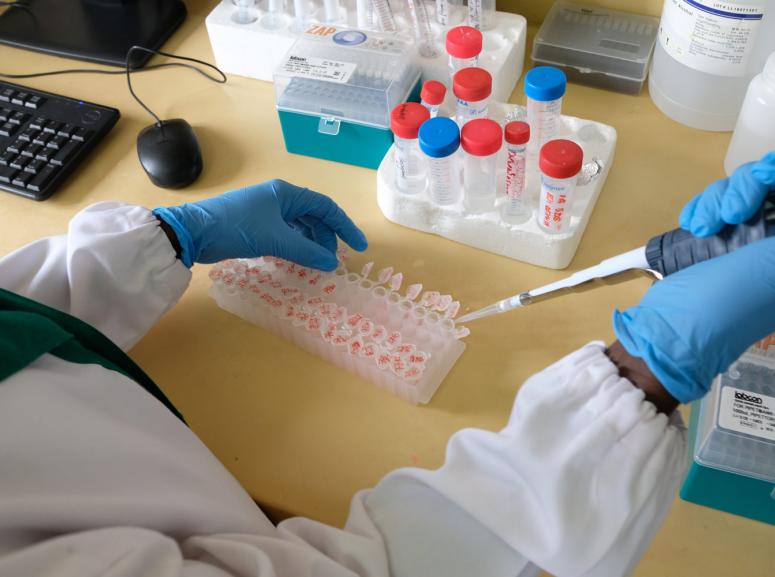
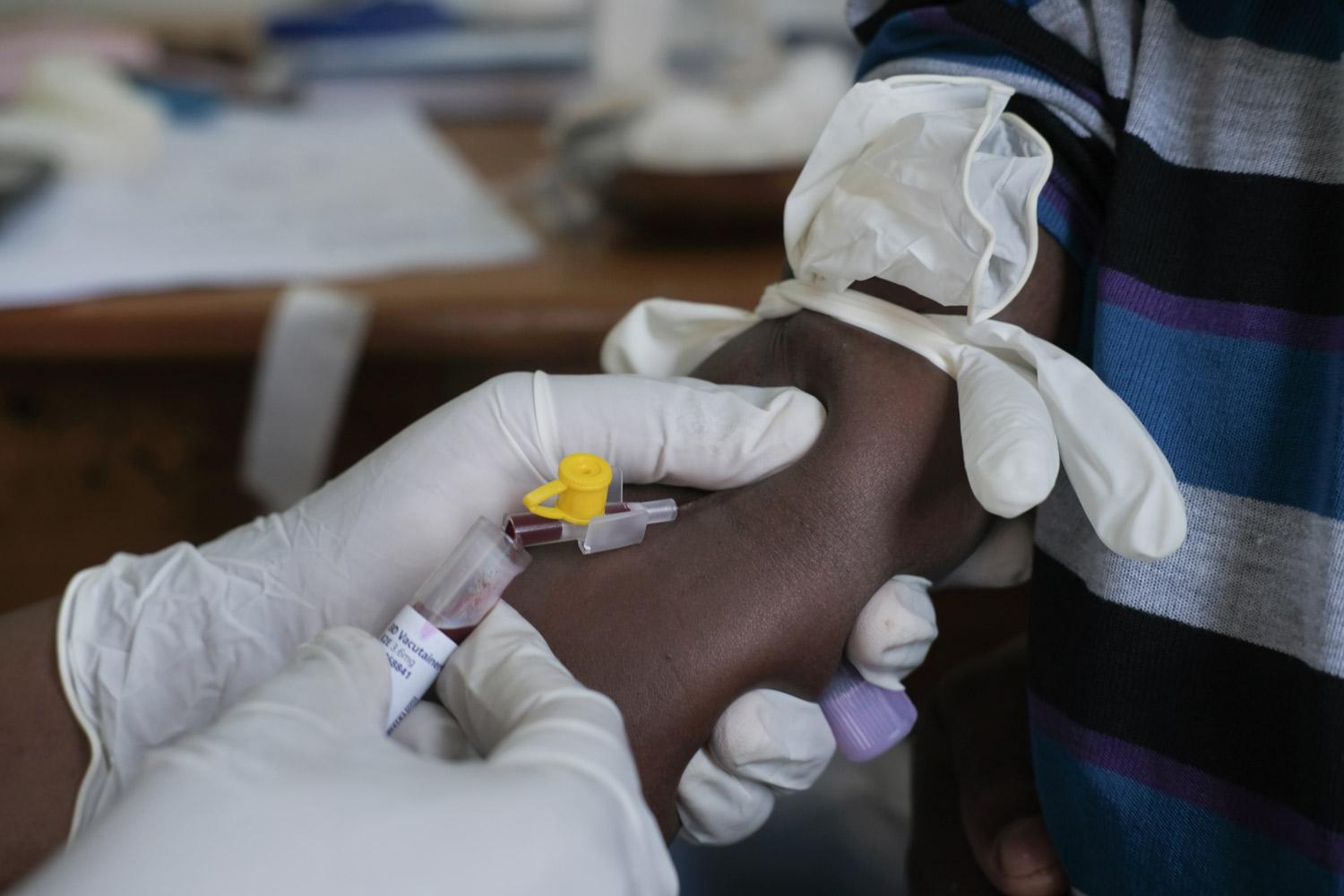
Epicentre Uganda benefits from a laboratory located within the Epicentre facility with a biosafety level 3. The laboratory is equipped to do parasitology, (with extensive experience in malaria microscopy), mycobacteriology (biosafety level 3 to perform cultures), microbiology (biosafety level 2, molecular biology; classical PCR and real-time qPCR, use of GeneXpert), biochemistry and haematology with skills to handle specimens for shipment according to IATA regulations. It also has a cold chain capacity for – 20 and – 80 degrees storage.
As of February 2021, the center is accredited to perform PCR testing for SARS-CoV-2 and thus supports the Ugandan Ministry of Health in the fight against COVID-19.
Education
Taking advantage of its position at the heart of the Mbarara campus the centre is also actively engaged in teaching at the MUST Faculty of Medicine. Research Centre staff contribute to teaching in research methods, supervision and mentorship in research to Faculty of medicine MUST students. In addition, Epicentre laboratory staff contribute modules to bachelor's and master's courses in microbiology, molecular biology, and medical laboratory sciences. Laboratory Science students have the opportunity to gain valuable experience in the Research Center’s laboratory.
Members of the Mbarara Research Center
Introducing Epicentre Uganda
Activities in Uganda
Workshop TB 2022
Find out more
Scientific Day Uganda 2017
Find out more
Yellow fever
Find out more
Tuberculosis
Find out more