Tuberculosis
According to WHO's predictive modeling, the number of people developing and dying from the disease could be much higher in 2021 and 2022. This is because the number of people diagnosed, treated, or receiving preventive treatment for the disease has decreased compared to 2019, and overall spending on essential TB services has decreased.
Challenges include under-diagnosis, as many people are unaware that they have the disease, and the emergence of treatment-resistant forms.
Tuberculosis (TB) is caused by Mycobacterium tuberculosis, a bacteria most commonly affecting the lungs. Although tuberculosis is largely preventable and curable, by 2015, tuberculosis became the world’s leading infectious killer. In 2016, 10.4 million people fell ill with TB, and 1.7 million died from the disease (WHO Global Tuberculosis Report). Almost one quarter of the world's population has latent tuberculosis infection and could develop the disease. In sub-Saharan Africa, the burden of TB is mostly driven by the HIV epidemic.
It’s been said that tuberculosis is a disease of poverty, as 95% of TB deaths occur in low- and middle-income countries. In particular children, HIV-infected persons, people suffering from malnutrition and prisoners, are at risk of TB. This may be due to lack of access to adequate diagnosis and treatment or to poor response to treatment. In addition, the emergence of resistance to two major anti-tuberculosis drugs, isoniazid and rifampicin (Multidrug resistant tuberculosis or MDRTB), is posing a major problem in tuberculosis control.
Are the latest innovations effective in vulnerable populations?
In the last few years there has been progresses in the diagnosis and treatment of TB. However, many of these new tests and treatment regimens have not been evaluated on the populations that could benefit the most. Epicentre works with Médecins Sans Frontières (MSF) measuring the importance of the disease in vulnerable populations, evaluating the implementation of new drugs, novel regimens, new diagnosis tests, and novel case management strategies in order to improve the patients’ outcomes.
Diagnosis remains a challenge
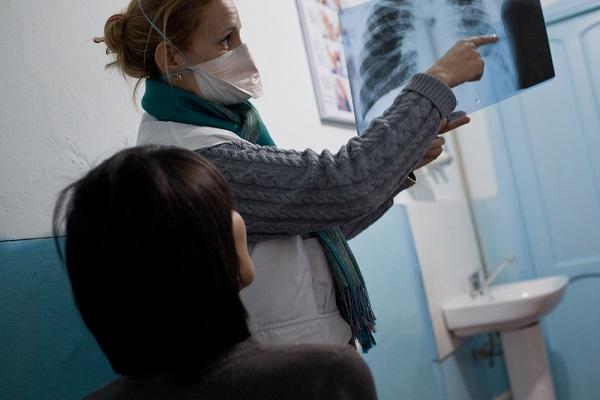
TB diagnosis remains a challenge in resource constrained settings, especially in children and advanced HIV-infected patients.
Epicentre has conducted several observational studies on the accuracy, the diagnostic value, the cost-effectiveness and the feasibility of using the point-of care urine based Determine TB-LAM (commercialized by Alere/Abbott), in HIV-infected patients in Kenya, Mozambique, Malawi and DRC. The same test has also been evaluated in severely malnourished children in Niger and in severely ill children in Uganda. The test has been found to have modest sensitivity but important diagnostic value for TB in HIV-positive patients. It is also feasible to implement in low resources settings and highly cost-effective.
Epicentre is currently conducting a study to evaluate a new point-of-care urine-based TB-LAM test (developed by Fujifilm and not commercialized yet), the FujiLAM, in HIV-positive adults. Funded by the French National Agency for AIDS Research (ANRS) and viral hepatitis and MSF, the study is taking place in four sites with high HIV and TB prevalence: Uganda, Kenya, Mozambique and South Africa. First results indicate a high FujiLAM testsensitivity in HIV-positive TB patients and higher than that observed with, the currently recommended test Determine TB-LAM (Alere/Abott). The FujiLAM test is also feasible to implement, well accepted by test users and by patients.
New therapeutic approaches
TB-speed: diagnosing to stop TB in children
Despite efforts, TB remains a leading cause of death in children under 5 years of age. About 1.2 million new cases were diagnosed worldwide in 2018 and 230,000 deaths were expected in 2019. In 2020, 1.1 million children fell ill with TB globally.
A majority of these children die without access to treatment and most often because they were not diagnosed. The TB-Speed project coordinated by the University of Bordeaux aims to reduce child mortality. Epicentre, through the Mbarara Research Center in Uganda, is one of the technical partners of the study.
One of the objectives of TB-speed project is to address the lack of a rapid, effective and easy-to-use diagnostic test for tuberculosis in children. The four-year project is taking place in seven countries in Africa and Southeast Asia (Cameroon, Côte d'Ivoire, Mozambique, Uganda, Sierra Leone, Zambia, and Cambodia) and is expected to help screen more than 77,000 children.
More effective and better tolerated treatments
Epicentre also participates in international therapeutic trials with its research Centre in Mbarara, (Uganda). The STATIS ANRS (French National Agency for Research on AIDS) trial compared the mortality of an intensive, systematic, and continuous screening for TB and the use of systematic empiric TB therapy in advanced HIV-infected adults. The first study results showed that systematic TB treatment is no superior to intensive TB screening in this population.
Shorter treatment to improve adherence
Together with the St Georges University London, Epicentre is involved in several trials evaluating shorter anti-TB regimen using higher dose of rifampicin. The current treatment involves daily medication for 6 months. If treatment could be reduced to 3 or even 4 months, it would improve compliance and daily life for patients while limiting the emergence of drug resistance. After several previous studies, the international Rifashort study is evaluating the efficacy and safety of a 4-month regimen using double and triple doses of rifampicin.
Datura: more intense treatment in HIV-positive patients with tuberculosis
Funded by EDCTP and ANRS, the DATURA trial - Determination of Adequate Tuberculosis Regimen in Adults and adolescents hospitalized with HIV-associated severe immune suppression - aims to assess whether more intensive initial treatment of tuberculosis, consisting of increased doses of the main antibiotics used, rifampicin and isoniazid plus corticosteroids, increases the chances of survival of hospitalized adults and adolescents co-infected with HIV and tuberculosis, as compared to standard treatment of TB. The Mbarara Research Center is one of the study sites.
Promoting household TB contacts’ screening and preventive treatment coverage
The Mbarara centre also took part in the CONTACT study coordinated by IRD, which is part of the the Elizabeth Glaser Pediatric AIDS Foundation (EGPAF)’s CaP-Tuberculosis (TB) Project and was funded by global health organization Unitaid.
This is the first cluster randomized controlled trial to evaluate a community-based intervention for tuberculosis screening and TPT management of household child contacts by community-health workers.
The study compared its effectiveness, feasibility, and cost-effectiveness to the facility-based standard of care. The study was performed in Cameroon and Uganda and shows that community-based contact investigations performed by community-health workers increases the coverage, initiation, and completion of preventive treatment of tuberculosis (TPT) among child contacts under 5 years old or 5-14 years old children living with HIV. The proportion of child contacts of all ages who were screened for TB increased from 47.3% in the standard of care arm to 81.9% in the intervention arm and the proportion of child contacts in the TPT target group that initiated and completed TPT increased from 61.7% in the standard of care arm to 79.9% in the intervention arm.
The intervention prevented 15 tuberculosis deaths in Cameroon and 10 in Uganda. The incremental cost-effectiveness ratio was $620 per disability-adjusted life-year (DALY) averted in Cameroon and $970/DALY averted in Uganda.
The CONTACT study confirms that decentralizing contact investigation at the household level using community-health workers is feasible and effective in improving child contact screening coverage, TPT uptake and completion as compared to facility-based standard of care.
Drug-resistant TB is on the rise
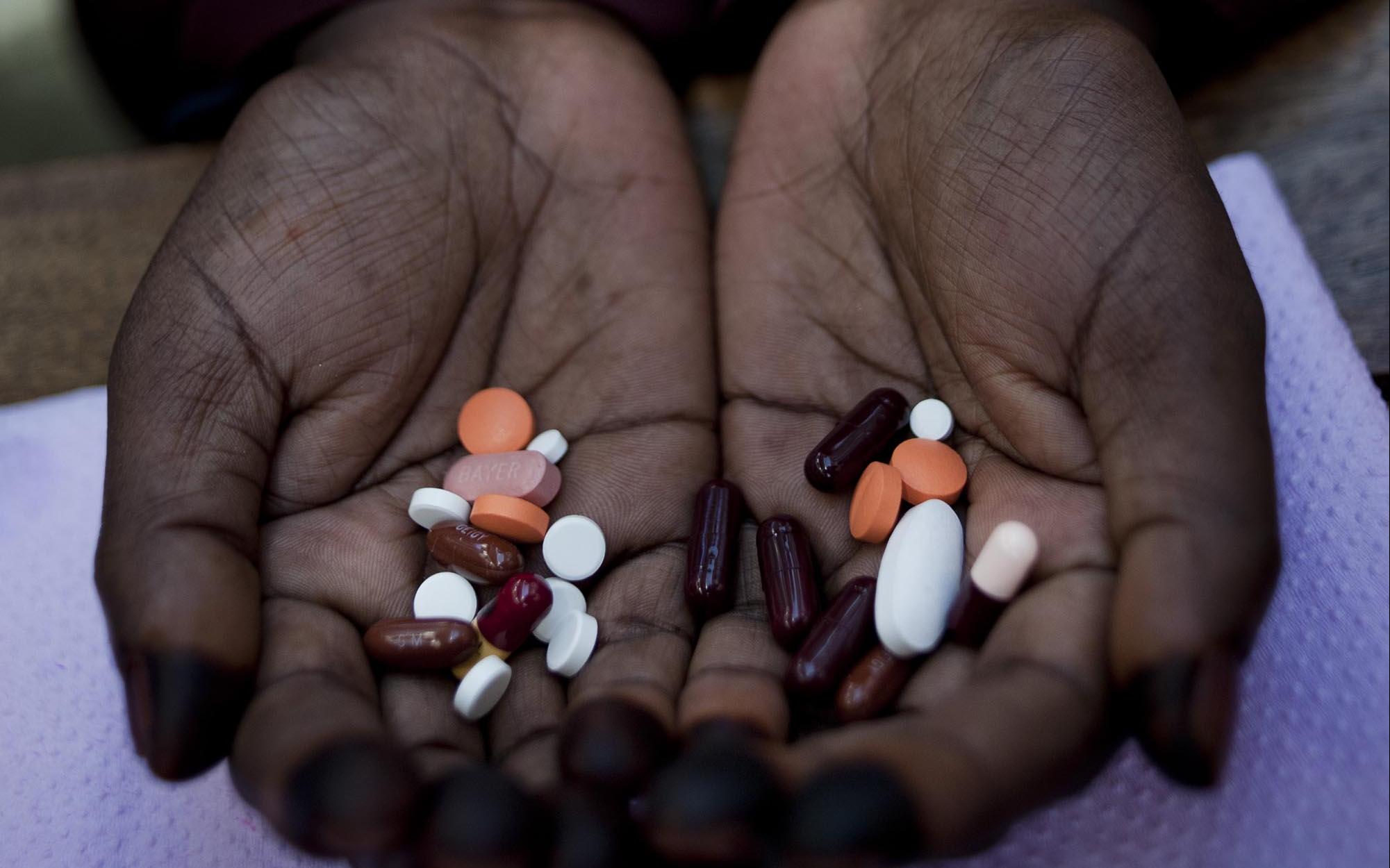
The emergence of resistance to anti-tuberculosis drugs is a global threat to the fight against tuberculosis. Resistance to one or more antibiotics is already observed and more worryingly, more and more multidrug-resistant strains, i.e., those that have become insensitive to several drugs, including at least the two most effective (isoniazid and rifampicin), are emerging.
In 2018, according to WHO, 484,000 cases of resistance to rifampicin, the most effective first-line drug, were diagnosed globally, of which 80% were multidrug-resistant TB (MDR-TB).
EndTB: shorter, less toxic and more effective treatments for "multidrug-resistant tuberculosis" (MDR-TB).
EndTB is a large project which took place in 17 countries and involved Partners In Health, Médecins Sans Frontières, Interactive Research & Development. Funded by UNITAID, it is organized around an observational study and 2 clinical trials sponsored by MSF and in which Epicentre, Harvard Medical School and the Institute of Tropical Medicine in Antwerp are involved.
Current treatments for multidrug-resistant tuberculosis can last up to 24 months. Only 54% of them are successful and they are often accompanied by significant side effects. For some patients, they result in the ingestion of 14,000 pills and painful daily injections for months. The cost, difficulty and duration of these treatments make them difficult to implement.
The trial found three new drug regimens that can deliver similar efficacy and safety to conventional treatments while reducing treatment time by up to two-thirds. The endTB clinical trial evaluated five experimental regimens for MDR/RR-TB against the standard of care in two distinct analysis populations. endTB regimens 1, 2, 3, demonstrated non-inferiority to the control in both primary analysis populations, establishing their success in treating RR-TB. Regimens 1, 2, and 3 achieved favorable outcomes in 89.0%, 90.4%, and 85.2% of participants, respectively. In addition, the trial supports the use of a fourth regimen as an alternative for people who cannot tolerate bedaquiline or linezolid; at least one of these two drugs is in every current World Health Organization-recommended regimen for MDR-TB.
The observational study in which Helena Huerga, an epidemiologist at Epicentre, is one of the principal investigators aims to collect data on the safety and efficacy of protocols using the drugs authorized in 2012 and 2013, respectively, bedaquiline and delamanid, for multidrug-resistant tuberculosis. The interim results show these two molecules to be safer than the drugs currently in use and suggest an improved response to treatment of resistant forms of TB. However, these results are not homogeneous across countries.
TB and Hepatitis C
Tuberculosis and Hepatitis C are infections that have similar high-risk populations and therefore are it is common that these infections are concomitant. In addition, hepatotoxicity is common during MDR-TB treatment and Hepatitis C is also associated with increased toxicity. MSF is systematically screening for Hepatitis C all patients starting treatment for MDR-TB in Armenia and provides treatment with new direct-acting antivirals (DAAs) to those with active infection. Epicentre is conducting a study aiming to assess the safety, effectiveness, and feasibility of this pioneering program.